
Localization


This article is written from the perspective of a language service provider (LSP). It is intended for manufacturers of life sciences equipment and medical devices who have grown to the point where they export their products outside of their domestic markets. They may have established subsidiary offices overseas or simply use local distributors. It is assumed that the manufacturer has a translation management system (TMS) and has selected at least one LSP who can translate source documents into all the required target languages.
Attention is drawn to the role of the subject matter expert (SME) and potential bottlenecks which can occur in the later stages of the localization process.

Pricing is sometimes agreed upon by an LSP or translator team before actually translating a live project through to the final proofreading stage. If the correct pricing is not established from the onset, problems, including loss of translation resources, could occur down the line. Therefore, establishing an efficient workflow and figuring out potential bottlenecks* before agreeing on pricing is essential for both the manufacturer and their LSP.
Simple, four-station workflow for the translation and DTP of marketing materials.
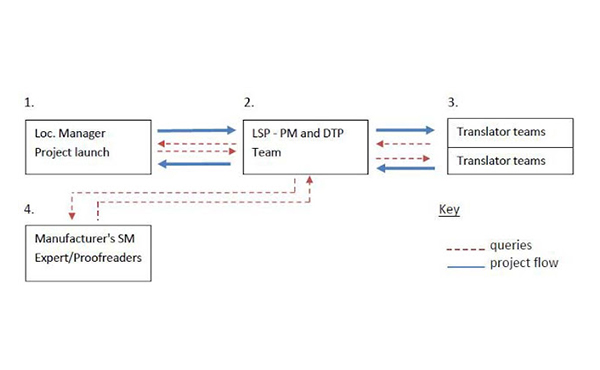
Let us take the example of a manufacturer’s Localization Manager with a 5 page marketing document describing a spectro analysis product and its benefits. The source document needs to be translated into 4 Asian languages with a finished PDF brochure as the final delivery.
In this stage, the manufacturer sends the translation kit to the LSP. After asking initial questions, the project is sent to the four translation teams. The teams then submit any questions they have for the manufacturer’s SME. This is where delays often occur since no progress can be made until the SME provides the necessary feedback.
Once translation teams submit their work, it is published in PDF format. The PDF is submitted to the manufacturer’s SMEs for the first round of proofreading. Changes are made and the document is sent to the translation teams to implement or alter the revisions. The PDF is sent to the SMEs for a second round of proofreading. If the second pass is accepted, it is sent to the localization manager to be finalized. The translation memory is then updated by either the manufacturer’s localization department or by the LSP.
Note: The number of necessary proofreading rounds has the potential to increase project management costs and upset translators. Pricing should be based on two passes from the start to avoid unexpected margin erosion and unhappy translator teams.
The easiest parts of the production process are desktop publishing (DTP) and translation! The real challenge often lies with the SMEs. If the manufacturer has dedicated SMEs who understand the importance of fast turnarounds, then delays are minimized. In some cases, however, SMEs in subsidiary offices or distributors have other responsibilities and view their proofreading duties as a second to their everyday job. If the manufacturer fails to ensure its SMEs give priority to the proofreading of projects (text and DTP), delays will occur and will often extends a project’s completion time.
Another issue is finding appropriate SMEs. Depending on the level of technology and sophistication of the product being localized, internal product experts or external PHDs and professors of, say biochemistry, might be needed. Therefore, the manufacturer has to make sure either she/he has the internal SME resources or commissions the LSP in good time to find appropriate external subject matter experts.

Localizing marketing materials for high-tech products often involves more stages than, for example, those needed for food & beverages. Firstly, translators with the appropriate education and experience need to be selected. Individuals will need to be tested, graded, and have their output scored. If an LSP, for example, has been selected to translate into 15 languages, then 15 translator teams or an appropriate number of partner language vendors will need to be managed. LSPs should only agree to work on projects they feel they are equipped to handle.
The LSP should talk in depth with the manufacturer to discover their expectations, required processes, and gain insight about the SMEs, such as where they are based, what their daily responsibilities are, and what their likely proofreading capacities are. Only when translation capacities and SME proofreading capacities have been established can delivery times and volumes be determined.
Should the manufacturer require the LSP to source external SMEs, a budget and schedule need to be established and agreed upon. The more high-tech and sophisticated the manufacturer’s products are, the more difficult it will usually be to find the right SMEs.
Transparent, comprehensible, and easy-to-maintain information on the project’s process needs to be provided by the LSP for the manufacturer. This information could be provided through a translation management system or other means. It is the responsibility of the LSP to proactively alert the manufacturer to both problems and suggested improvements once indentified.
The above is just an abridged version of a complicated localization process and highlighted areas I think would be of interest.
We would recommend choosing an experienced LSP in the required subject matter, that can take care of many of the services needed in Life Sciences, Healthcare, and Medical Device, has the required languages, can facilitate the manufacturer’s documentation processes, and has a high level of integrity and trust.
Reach Out Today! Talk To Our Experts Now! or Request A Free Quote
Hansem Global is an ISO Certified and globally recognized language service provider. Since 1990, Hansem Global has been a leading language service company in Asia and helping the world’s top companies to excel in the global marketplace. Thanks to the local production centers in Asia along with a solid global language network, Hansem Global offers a full list of major languages in the world. Contact us for your language needs!
 Transforming B2C Sales Training with Gamification
04.14.2024
Transforming B2C Sales Training with Gamification
04.14.2024
 The Impact of Localization on Salesforce’s Success with Hansem Global
04.07.2024
The Impact of Localization on Salesforce’s Success with Hansem Global
04.07.2024
 Mastering Right-to-Left (RTL) Language Localization: Avoid the Top 5 Desktop Publishing (DTP) Mistakes for Global Success
04.03.2024
Mastering Right-to-Left (RTL) Language Localization: Avoid the Top 5 Desktop Publishing (DTP) Mistakes for Global Success
04.03.2024